SUNOSI
Regulated Healthcare Platform | Compliance-Driven Interface Documentation & Workflow Analysis
Managed structured documentation and compliance analysis for clinical data systems in a regulated healthcare environment. Translated complex datasets and regulatory requirements into consistent, audit‑ready documentation across adverse‑event matrices, safety hierarchies, and form‑validation workflows.
Disclaimer: The work shown was completed during full-time employment and is shared to highlight my role and contributions. This is not a client of Stone Interface.
Project Context
Documentation and analysis of data-heavy interface modules within a regulated clinical environment, including hierarchical safety structures and form-driven workflows. Focusing on structured output accuracy, consistent component behavior, and strict adherence to defined regulatory and system constraints.
Scope of Work
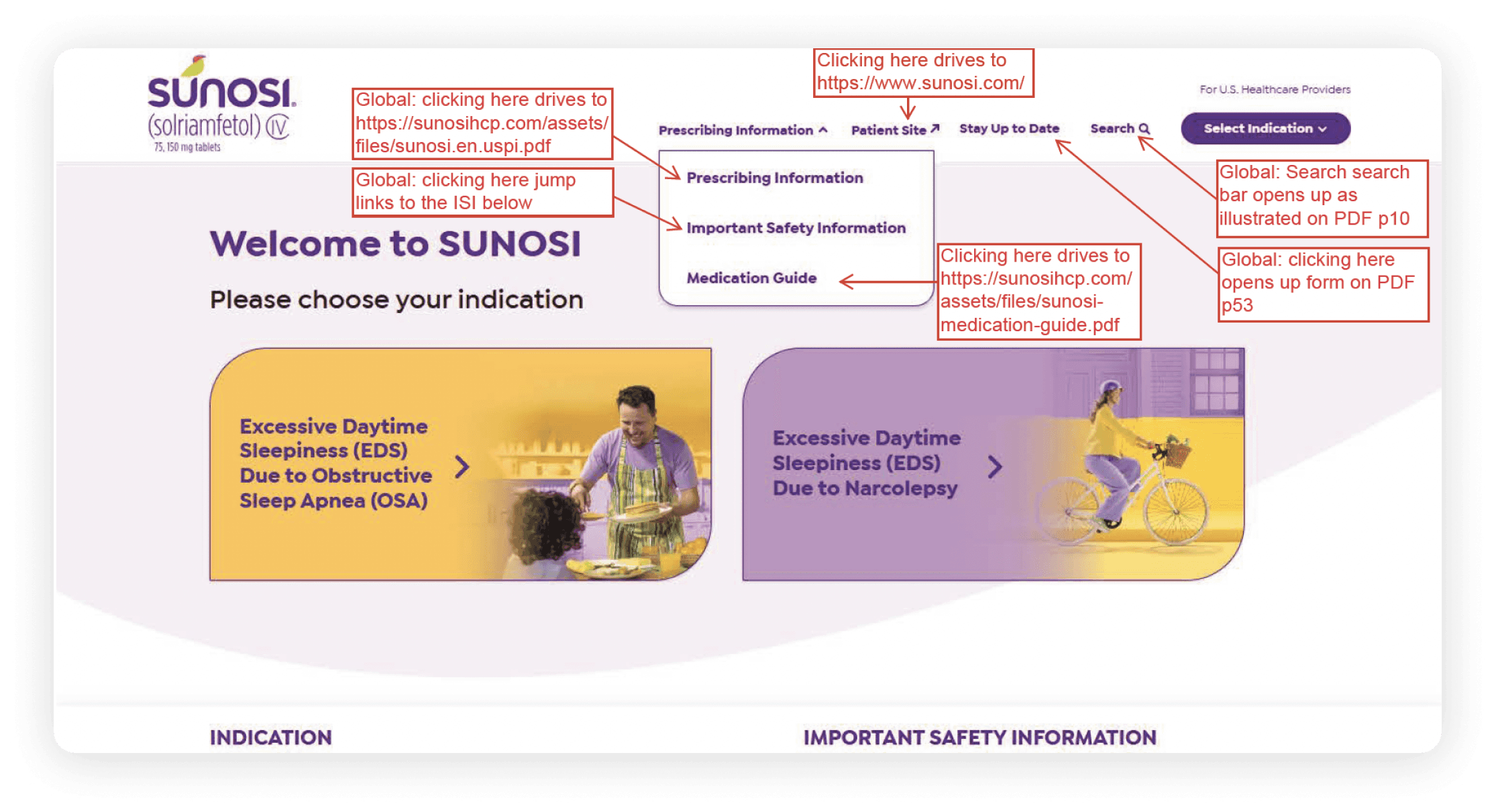
Global navigation link mapping and destination documentation
Source citation traceability across regulated clinical content
Required vs. optional field logic and error state specification
Form validation logic & controlled input states
Compliance Remediation with WCAG 2.1 AA and internal Pharma safety standards
Structured large-scale adverse event reporting tables supporting standardized comparison workflows across multiple dosage conditions and placebo datasets. Organized dense clinical reporting information into scalable, validation-oriented layouts optimized for operational review and regulatory consistency.
Worked closely with UX architects and copywriters to produce functional specifications documenting global navigation behavior, link‑destination mapping, and content‑level source traceability for a regulated clinical platform. Work included mapping exact URLs, documenting PDF and ISI anchor behavior, and maintaining citation links from each claim to its precise source. Modal hierarchies were captured using full-disclosure sequencing and component‑level references.


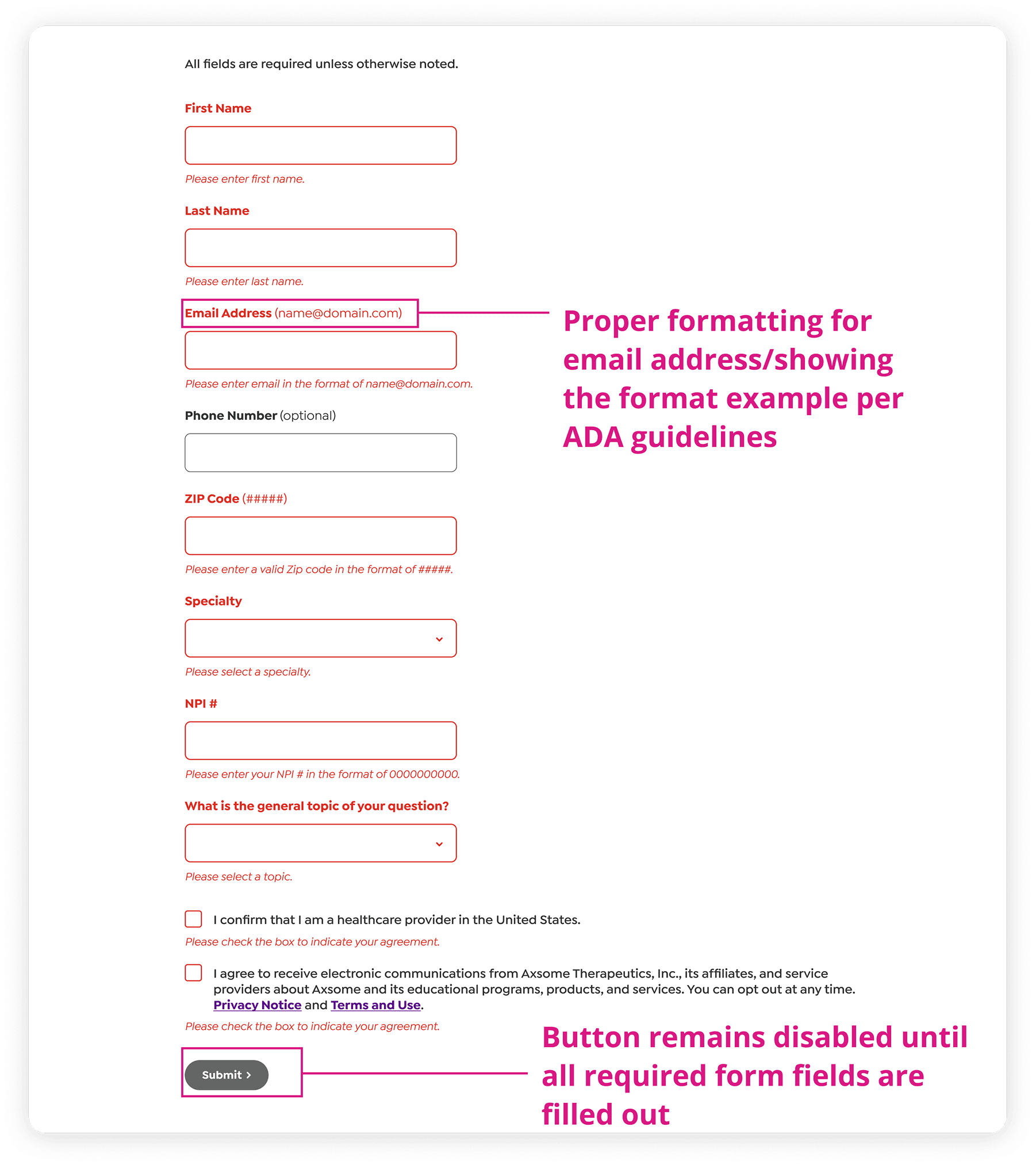
Documented field‑level validation rules and ADA‑compliant formatting for a regulated healthcare data‑capture form. Specified input formats, error‑state messaging, required/optional logic, and disabled‑state behavior to ensure all controlled inputs met accessibility and compliance requirements.
Utilized WCAG 2.1 AA/AAA guidance in collaboration with ADA specialists to remediate contrast, typography, and state visibility, ensuring high-stakes interfaces remained compliant and functional within a strictly regulated operational environment.